
 Electronegativity is the attraction of an atom for electrons.
Electronegativity is the attraction of an atom for electrons.
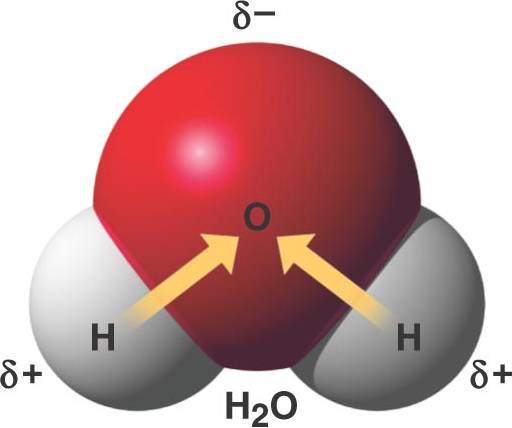
A water molecule is polar because oxygen is more electronegative than hydrogen.
There is a partial negative charge on the oxygen and partial positive charges on the hydrogen atoms.
The unequal sharing of electrons results in polar covalent bonds.